Temperature
Temperature is defined as the degree of hotness or coldness of a substance. The atmosphere is warmed only slightly (0.5 to 1 degree F. per day) by direct—mainly shortwave—solar radiation. Most of the warming develops through conduction and convection from the heated surface of the earth, and from long wave radiation from the surface.
You will see later that temperature has far reaching effects on general atmospheric circulation, the formation and movement of air masses, and regional weather patterns. These are all important elements of fire weather. But with fire weather we are also concerned with smaller scale patterns —those which change from hour-to-hour, from one slope aspect to another, from one forest fuel to another, from a closed canopy to a forest opening, etc. In these smaller scale patterns, temperature variations are often the controlling factor.
Sampling the weather at regular weather observation stations commonly identifies large-scale weather patterns. Small-scale patterns with their many variations cannot be derived from measurements taken at usually widely spaced weather recording stations. Sometimes portable instruments permit satisfactory measurements, but more frequently, local variations must be identified by judgment based on technical knowledge of how surface materials and the air around them is heated and cooled, and how thermal energy is transferred between the earth’s surface and the lower air.
The following text discusses focuses on the variations in surface and air temperatures, and why they occur.
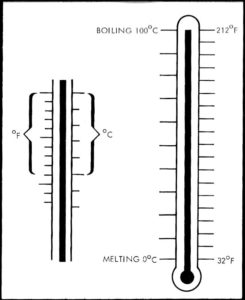
The two common temperature scales in use are Fahrenheit and Celsius. Nine degrees Fahrenheit equals five degrees Celsius.
A thermometer embedded in a solid or immersed in a liquid soon comes to temperature equilibrium with the substance and indicates the actual temperature of the substance. Measuring the air temperature is more difficult. For example, during the daytime if sunlight strikes the bulb of the thermometer, the reading will be higher than the air temperature because of direct radiation. At night, if the bulb is exposed to the sky, the reading will be influenced by the outgoing radiation of the bulb, and will be lower than the air temperature. So most thermometers are shielded from radiation to increase their accuracy by minimizing the error factor produced by radiant heat, leaving only conduction heating to confuse the actual air temperature reading. A standard instrument provides such shielding at fixed locations while permitting free flow of air past the thermometer inside. A hand-held thermometer should first be “shaken down” and placed in a shaded area to ensure the more accurate reading.
Representative Measurements
For optimum use in fire control, the measured air temperature at a fire weather station should be representative of the surrounding conditions. As you will see, many factors affect the air temperature including the type of ground surface, nearby buildings or trees, local topography, and the distance from the ground of the temperature-measuring device.
To ensure comparability between the fire weather stations, some standards were developed. Measurements are recorded at a standard height of 4-1/2 feet above the ground. Locations near buildings or other obstructions are avoided, as are types of ground surfaces such as concrete or asphalt that could dramatically affect temperature readings. Purely local effects are avoided where possible.
The local variations in temperature that are lacking when readings are used for fire weather forecasting or for area fire-danger ratings become most important when judgments concerning fire behavior at a specific time and place must be made. Then it is necessary either to obtain closely spaced measurements to reveal the temperature variations, or to make judgments based on personal knowledge of where and how these variations might occur.
The causes of these temperature changes are many and varied, but three very important processes underline all causes:
- The heating and cooling of the earth’s surface due to radiation
- The exchange of heat between the surface and the air above it
- The conversion of thermal energy in the atmosphere to other forms of energy, and vice-versa
All three processes vary continuously.
Earth Surface Temperatures
Effects of Solar Radiation
The temperatures of most materials that comprise the surface of the earth (except for water and ice) have a wider temperature range than does air. The temperature of surface materials is especially important because air is heated and cooled primarily by contact with heated or cooled surfaces.
Some factors affect surface temperatures because they influence the amount of solar radiation that strikes the surface or because they cause the earth’s radiant heat to become trapped.
A lower sun angle results in the reception of less solar radiation per unit area, and a lower surface temperature. More hours of daylight transfers into more solar heating and higher surface temperatures. Conversely, more hours of darkness result in more cooling and lower surface temperatures.
Topography also plays an important role in local surface temperature variations. Differences in topography cause local variations in the angle at which the sun’s radiation strikes the ground surface. Both the steepness and the aspect of a slope affect surface heating and cooling. Surfaces more nearly perpendicular to incoming radiation receive more heat per unit area than do those more nearly parallel to incoming radiation. As the sun progresses across the sky, its rays are more nearly perpendicular to individual slopes and aspects at varying times.
South-facing slopes, which in our Northern Hemisphere collect more nearly direct rays from the sun during most of the day than do north-facing slopes, may have a surface temperature in midsummer as high as 175 degrees F. Level surfaces reach their maximum temperatures around noon, but the maximum temperature on a slope depends then upon the inclination, and the orientation of the slope, and the time of day. Accordingly, east-facing slopes reach their maximum temperature rather early in the day; west-facing slopes attain their maximum temperatures in the late afternoon.
In general, the highest surface temperatures are found on slopes facing to the southwest.
Shading and scattering by any means including clouds, smoke or haze in the air, or objects such as trees, reduce the solar radiation reaching the ground surface. In hilly or mountainous regions, higher ridges shield lower elevation surfaces from incoming radiation thus reducing the hours of sunshine.
All vegetation creates some shade, but the variations in type and density cause local differences in surface temperatures. In open stands of timber, the temperature changes throughout the day for shaded versus sunlit areas according to the position of the sun. Surface temperatures respond quickly to these changes. For example, a forest floor with a mottled sun and shade pattern may have temperature variations during the summer of as much as 50 to 60 degrees F. within just a few feet.
UPPER LEFT: Surfaces more nearly perpendicular to incoming radiation receive more heat per unit area and become warmer than those that are more nearly parallel to the incoming radiation.
LOWER LEFT: As the sun arcs across the sky, its rays are more nearly perpendicular to varying slopes and aspects at different hours of the day. South-facing slopes receive more nearly direct rays than do north-facing slopes.
UPPER RIGHT: In open strands of timber, surface temperatures vary considerably from shaded to sunlit areas.
LOWER RIGHT: Clouds both absorb and reflect incoming radiation, thereby reducing surface temperatures.
Both liquid water droplets in clouds and the water vapor in the atmosphere directly affect surface temperatures. They each absorb some incoming radiation, and clouds reflect much of the solar radiation. The thicker and lower the clouds, the less incoming radiation strikes the surface. Surface temperatures may drop as much as 50 degrees F. within three minutes when a thick cloud passes overhead in clear midsummer weather. Water droplets in clouds, and invisible water vapor in the air also influence the surface’s cooling at night. Both absorb much of the outgoing thermal radiation that then is reflected back to the earth. Thus surface temperatures are usually much lower on clear nights than cloudy ones. This lack of water vapor in the air is one reason why surface temperatures in the desert drop so low at night.
Like clouds, a blanket of smoke from a forest fire can result in significantly lower daytime surface temperatures and higher nighttime temperatures than when skies are otherwise clear.
Effects of Surface Properties
When radiation strikes a surface, there are several properties of the surface substance that affect its resulting temperature.
First is the capacity of a substance to absorb or reflect radiation. Dark materials generally absorb most of the radiation in the visible wavelengths, whereas light materials reflect most of this radiation back to space. Since dark soils and forest litter are rather good absorbers (and poor reflectors) of radiation, they will become hotter than light-colored soils. Dark pavement also becomes quite hot on a sunny day. The temperature of the tree crown in a forest will rise also, but not as much. Some of the incoming radiation is used in processes other than heating, such as in the production of food and in the vaporization of the moisture released by transpiration.
Substances that are good radiators of long wave radiation emit heat rapidly from their surfaces at night when exposed to a clear sky. If they are not supplied with heat from within, these surfaces become quite cold at night. Tree crowns, grass, plowed land and sand are all good radiators.
The absorptive and emissivity of a surface varies with the wavelength of the radiation and the temperature. But under identical wavelengths and temperature, absorptive and emissivity are assumed to be the same.
Snow is an interesting substance in that its properties are very different at different wavelengths. In the visible portion of the spectrum, snow will reflect 80 to 85 percent of the incoming shortwave radiation. This accounts for its white color. For long wave radiation, however, snow is an extremely good absorber and a near-perfect radiator. Therefore, a snow surface will warm little during the day, but cools by radiation extremely well at night. You will see later that these characteristics make snow ideal for the formation of cold, dry air masses.
A second property of surface materials affecting temperature is transparency. Water is primarily transparent to incoming radiation, while opaque materials are not. The heat absorbed by opaque substances is concentrated in a shallow surface layer, at least initially. But radiation penetrates deeply into water, heating a larger volume. This is one reason why opaque substances such as land become warmer during the day that water does. (This is not, however, the most important reason: The downward mixing of warmed surface water by turbulent motion is more important in distributing the incoming heat through a large volume.)
A third property is the conductivity of the substance. When incoming radiant energy strikes a good conductor, such as metal, it rapidly transmits the heat throughout the material, raising the temperature of the metal to a uniform level. The same radiant energy applied to a poor conductor tends to concentrate heat near the surface, raising the surface temperature higher than that of the interior.
For example, wood is a poor conductor and heat applied to it concentrates at the surface and only slowly penetrates to warm the interior. Leaf litter is another poor conductor. Common rocks, damp soil and water —although not as efficient conductors of heat as metals —are much better conductors than wood, other organic fuels or dry soils. Air is a very poor conductor, so porous substances such as duff or litter with many air spaces will impede the passage of surface heat to the soil below.
To summarize, the surfaces of poor conductors get hotter during the day and cooler at night than the surfaces of good conductors.
At night when surfaces cool by radiation, the surfaces of good conductors do not cool as fast as those of poor conductors as long as there is heat below to replenish that lost at the surface by radiation. For instance, a weathered board lying on bare, open ground may have visible frost where none has formed on the nearby ground.
Specific Heat
Different substances have different heat capacities, but the specific heat of a substance is the ratio of its heating capacity to that of water. The specific heat then, is another reason why the surface temperature of substances vary under similar conditions of incoming and outgoing radiation.
A substance with a low specific heat will warm up rapidly as heat is added to it, simply because it takes less heat to change its temperature. Water has a high specific heat, and its temperature changes 1 degree F. when 1 Btu of heat energy per pound is gained or lost.
Wood, which has about half the specific heat of water, changes about 2 degrees F. with a change of 1 Btu per pound. Materials like charcoal, ashes, sand, clay and stone change about 5 degrees F. Litter surfaces composed of dry leaves, needles and grass have low heat capacities, and mentioned above, are also poor heat conductors.
For these two reasons, direct solar radiation often heats litter surfaces to temperatures far above the temperature of the overlying air without heating the soil below.
Since water has a high specific heat and is a fairly good conductor, the surface temperatures of substances are greatly influenced by the presence of moisture. Moist surfaces, when compared with dry surfaces, will not reach as high temperatures in the day, or as low temperatures at night.
This is another reason why arid and simi-arid areas, when compared with moist regions, have both higher daytime surface temperatures and lower nighttime surface temperatures.
The presence of moisture is also important because of the heat used in evaporation and released in condensation. We have seen that while 1 Btu will raise the temperature of one pound of water 1 degree F., nearly 1,000 are required to evaporate one pound of water under normal conditions of pressure and temperature. Thus when water is evaporated from a surface, cooling takes place at the surface with a corresponding reduction in the surface temperature. This is yet another reason why surfaces of moist substances have lower daytime temperatures than dry substances. At night, if vapor condenses, an equally large amount of heat is released to warm the surface.
Effect of Wind
Strong daytime winds near the surface tend to prevent high surface temperatures. Transfer of heat between the surface and the air is improved by mixing, which carries heat away from the heated surfaces. This air movement also transports moisture, increasing evaporation from moist surfaces and thus resulting the temperature rise. At night, the effect of strong winds is to prevent low surface temperatures by mixing warmer air downward thus bringing it into contact with the surface, where some of the heat can be transferred to the ground by conduction. Therefore, wind has a moderating influence on surface temperatures.
Air Temperatures
The exchange of heat between the air and the surfaces over which it flows is the “master controller” of air temperatures.
This exchange is a continuous process that takes place everywhere at all times.
How Air Is Heated
Incoming solar radiation heats the air directly by only 0.5 to 1 degree F. per day, depending mostly on the amount of water vapor present. The remainder of the heating comes from below, most of it by conduction, through direct contact with the warmed surface of the earth.
The heated surface air becomes buoyant and is forced upward by cooler most dense air. This convection may distribute the heat through a depth of several thousand feet during the day.
The final depth through which heat from the surface is distributed through the atmosphere will be affected by the lapse rate of the air. If the lapse rate is stable through a deep layer, the daytime mixing and heating of the atmosphere will be confined to a fairly shallow layer of perhaps 1,000 to 2,000 feet, and the temperature of this layer will increase rapidly. If through a layer of air several thousand feet deep, the temperature lapse rate approaches the dry-adiabatic rate, heated air parcels will be carried to much greater heights. Then the heating and mixing take place throughout a deep layer, and the rise in air temperature near the ground will be less and slower. Thus, we can see that the characteristic air-mass temperature at several thousand feet above the surface is important in estimating the maximum temperature of air near the surface.
The general effect of wind on heating of the air is one of stability. Strong winds cause more turbulence and mixing so that heat is distributed through a deeper layer and the temperature rise of the air near the surface is less. The greatest temperature raises result from surface heat that is neither cooled nor mixed by light winds.
Another factor in the heating of air near the surface that should not be overlooked is the absorption of the earth’s long wave radiation by water vapor. Since most of the water vapor is concentrated in the lower layers of the atmosphere, these lower layers are heated by absorption of earth radiation as well as by conduction and convection.
How Air is Cooled
Air cools at night by the same heat transfer processes —conduction, radiation, and convection —as it heats during the day. The surface begins to cool first by radiation cooling the air in contact with it. On clear, calm nights, this is the primary method of cooling. It is primarily the surface air layer that is cooled while the air aloft may remain near daytime temperatures.
Water vapor and clouds also lose heat to the sky by their own radiation, but at a slower rate than the heat lost near the surface thorough clear, dry air. When clouds or significant water vapor is present, much of the outgoing radiation from below is intercepted and re-radiated back to the surface. Thus, the surface is cooled more slowly.
Winds at night also reduce the cooling of surface air by bringing down and mixing warmer air from above. This process does not slow the surface radiative cooling, but it does spread its effects on air temperature through a deeper layer. On the average, however, the difference between day and night air temperatures is much greater near the surface than it is aloft.
Effects of Forests on Temperature near the Ground
In all situations, vegetation moderates air temperature within the vegetative layer. This happens for several reasons.
First, it intercepts both incoming and outgoing radiation and, therefore, has a marked effect on ground temperature. Secondly, green foliage does not warm up as much as ground or dry litter. Also, the leaf surfaces exchange heat with air through a deeper, less restricted boundary layer. These effects result in less pronounced temperature changes with increasing distance from the ground.
In all vegetative cover, the temperature distribution depends upon the nature and density of the vegetation. With plants such as low brush, the leaves form a nearly continuous upper surface, and this surface acts as the effective ground surface. The maximum daytime temperatures and minimum nighttime temperatures are near the top of the brush or dense plant cover, although temperatures near the ground are not greatly different.
The crowns of trees in a heavy forest form a nearly continuous cover, thus the canopy becomes, in effect, the air contact surface. The highest daytime temperatures are found near the crown top, and the temperature will decrease gradually between this level and the ground. Maximum air temperatures near the crowns may be 18 to 20 degrees F. warmer than air temperature near the ground. Above the tree crowns, the temperature decreases fairly rapidly with height, although never as rapidly as over bare ground. This is true because the temperatures of the tree crown surfaces in contact with the air are lower than bare ground, and because the air circulation around these surfaces is better.
Less dense vegetation will permit more solar radiation to penetrate to the ground than will a dense cover. The degree of partial ground shading provided by less dense vegetation determines the air temperature distribution between the ground surface and the canopy top. It will range between that found over bare ground and that under a closed canopy.
Air in the crown region has higher daytime temperatures than air beneath the crowns. Nighttime temperatures in a dense timber stand tend to be the lowest near the tops of the crowns, where the principal radiation takes place.

Openings in a timber stand tend to act as chimneys under the conditions of strong daytime heating and light winds.
Forest air temperatures at a standard 4-1/2 foot height in the afternoon are likely to be 5 to 8 degrees cooler than the temperatures in nearby cleared areas. Openings in a moderate-to-dense timber stand may become warm air pockets during the day. These openings often act as natural chimneys and may accelerate the rate of burning of surface fires that are close enough to be influence by these “chimneys.”
Night temperatures in dense timber stands tend to be lowest near the top of the crown where the principal radiation takes place. Some cool air from the crowns sinks down to the ground surface, and there is some additional cooling at the surface by radiation to the cooling crowns. Sparse timber or other vegetation will merely decrease the strength of the inversion just above the ground surface.

